 The author requests only that this statementĪccompany all such copies. Such copies may be byĪny method, present or future. Move the mouse pointer over the image above, which shows a list of the X-ray coordinates for each non-hydrogen atom from a PDB file (1pso. Jmol icon appears on the screen Click on the Jmol icon. Click on Dash Home To open the Jmol Application, click on Dash home. 12.04 Jmol version 12.2.2 And Java version 7. Menampilkan label atom (simbol unsur) Memvisualkan pasangan elektron. Knowledge of Jmol script language, and a minimal amount of HTML, are required. To record this tutorial, I am using Ubuntu OS version. Tutorial ini dibagi menjadi beberapa bagian: Memvisualkan molekul dari file yang tersedia. Insert molecular views with buttons and descriptions using a text editor. Its use is to be for non-profit educational purposes. JmolShell Download a tutorial-development environment to create tutorials that will display in a web browser. Notice that this staggers the hydrogens on that atom relative to those on adjacent carbon atoms.Ĭopyright © 1998, 1999, 2000, 2003, 2007 by Daniel Often, in addition to installing the program. Copyright Notice: Jmol Tutorial Creator++ is the property and trademark of the developer Ian Morrill. Nevertheless, to avoid torsional strain the molecule bends into the so-called envelope conformation, in which one of the carbon atoms is bent out-of-plane. One of the great difficulties of using the various existing programs that assist VS is its installation. In this tutorial I show how to plot the Molecular Orbitals generated from softwares such as Gaussian, Quantum ESPRESSO, Turbomole, etc. Planar cyclopentane has almost-normal bond angles of 108°. 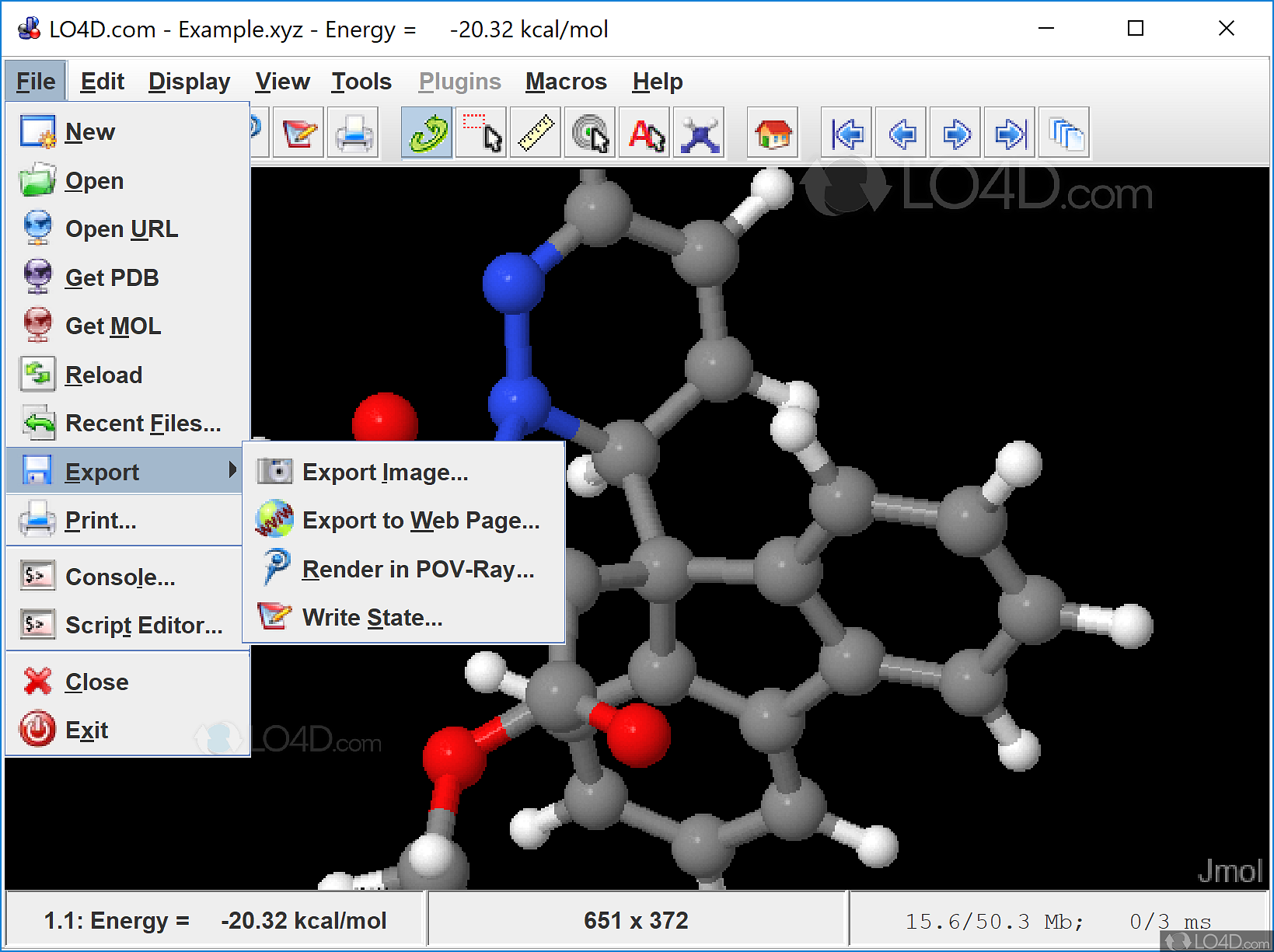
Cyclopropane and cyclobutane cannot avoid having large amounts of strain in their structures, but cyclopentane and cyclohexane can easily adopt conformations in which not only the angles have normal values but eclipsing (thus torsional strain) is minimized.Ĭyclobutane's lowest-energy conformer is slightly puckered - angle strain is increased in order to relieve torsional strain! 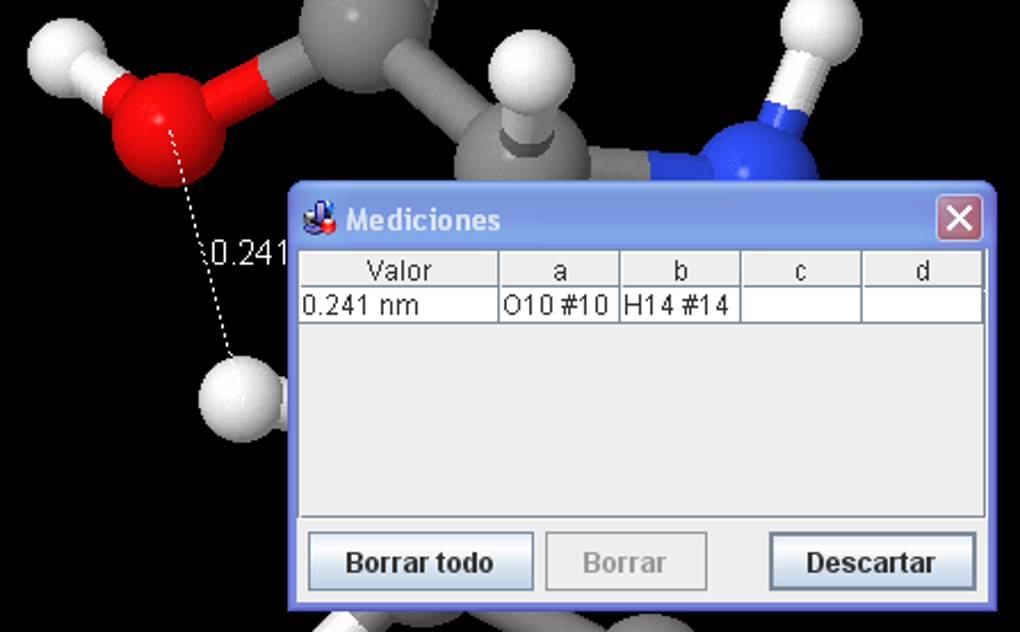
Serial Molecule Generator: transforming molecules by a sequence of user. Unsubstituted cycloalkanes, if planar, would not only have significant angle strain (caused by abnormal bond angles in the ring) but also considerable torsional strain (from eclipsing of adjacent hydrogens). Fragmenter: generating building blocks based on Recap rules from molecule libraries. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
